Neovascularization/Angiogenesis
![]()
PSI for Neovascularization/Angiogenesis
Neovascularization is the formation of new microvascular networks. In vivo, this process is primarily achieved through Angiogenesis, the sprouting of new vessels from existing vessels. Neovascularization/Angiogenesis plays a crucial role in many therapies and pathologies which is why it is so widely studied in many fields among them, Tissue Engineering and Regenerative Medicine, Vascular Biology, and Cancer Biology. Many models exist, depending on the application, for drug development/delivery, disease characterization, and stem cell treatment. There is a need for tools that can be used for assessment of new vessel formation, preferably through non-invasive and non-destructive methods. The PeriCam PSI Laser Speckle Contrast Imaging System can be used to track changes in microvascular blood perfusion overtime. Studies have shown that increases in perfusion correspond to more functional blood vessel formation. The PeriCam PSI has been used in several of these models.
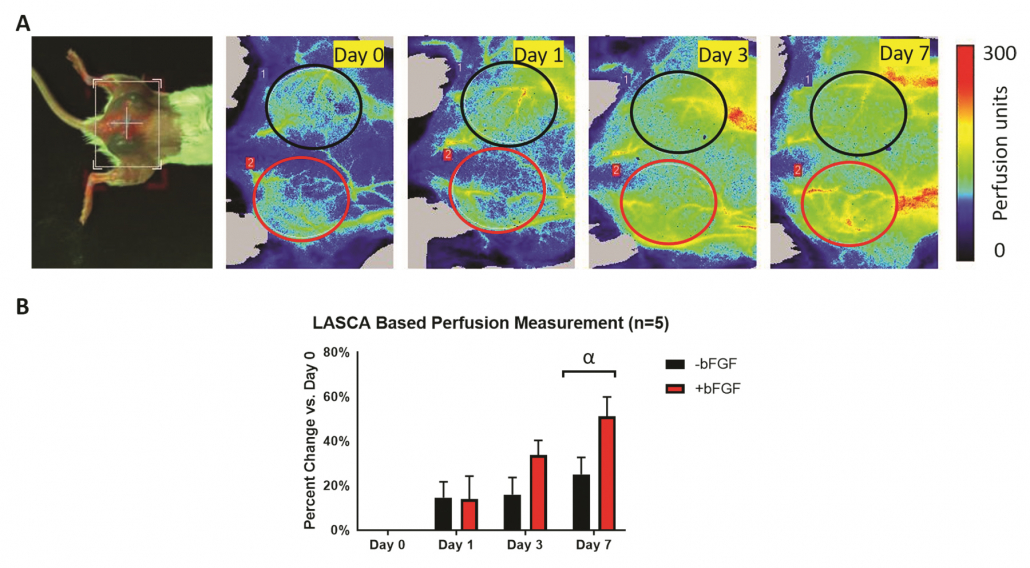
(A) Photo of dorsal view and longitudinal PSI Perfusion images of a mouse with two subcutaneous fibrin scaffold implants with and without angiogenic growth factor. The ROIs were chosen based on the physical location of the implants. (B) Quantification of the change in perfusion relative to day 0, based on an ROI analysis of the PSI Perfusion images, shows an overall increase in perfusion over time. The greatest change in perfusion was observed on day 7, with +bFGF scaffolds trending toward greater perfusion than -bFGF scaffolds 1-2.
Figure courtesy of Dr. Mario Fabiilli from LED-Based Photoacoustic Imaging for Monitoring Angiogenesis in Fibrin Scaffolds Author: Yunhao Zhu, Xiaofang Lu, Xiaoxiao Dong, et al Publication: Tissue Engineering Part C: Methods
Publisher: Mary Ann Liebert, Inc. Date: Sep 1, 2019.
Overview
PeriCam PSI has been proven to be a useful tool in a range of different fields within the area of neovascularization and angiogenesis research. One main field is the research of pro-angiogenic therapies for peripheral arterial disease and wound healing in pre-clinical animal models. For PAD, Ischemia is surgically induced in the lower extremity and confirmed using the PeriCam PSI and the dynamic process of perfusion recovery can be followed over time. Similarly, cutaneous wounds are created, and the PeriCam PSI is used to follow normal healing. In both models, the PeriCam PSI enables the researcher to follow the same subjects longitudinally saving time and money by reducing the number of animals needed for a study.
Another field is the study of cancer in the CAM Assay and Subcutaneous tumor models and the development of anti-angiogenic agents for treatment. The PeriCam PSI can be used to monitor perfusion in the tumors as they progress and assess the efficacy of therapeutics aimed to starve them.
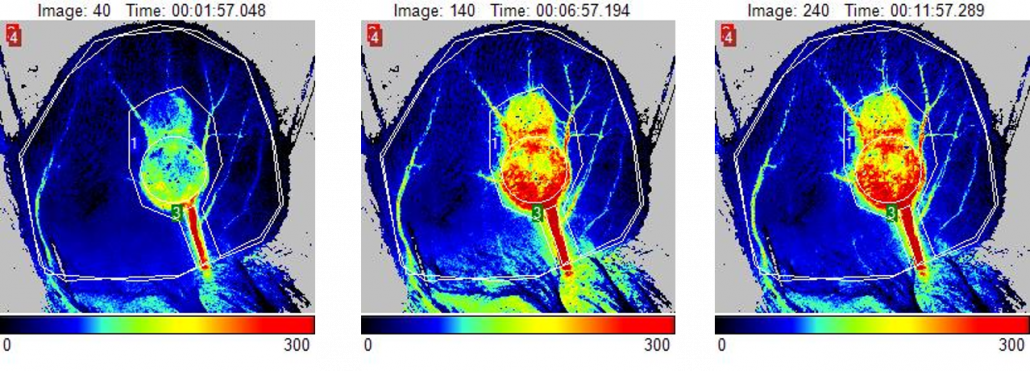
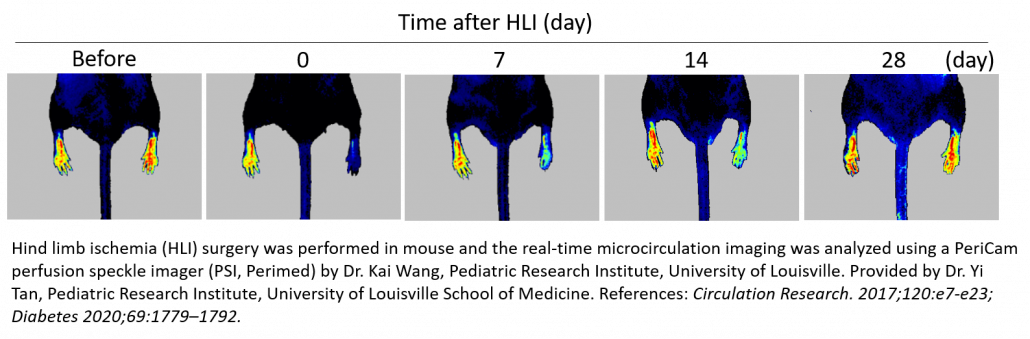
Hind Limb Ischemia (HLI)
The Murine Hindlimb Ischemia preparation is a commonly used and important model for studying peripheral arterial disease. It involves unilateral femoral artery ligation and excision to induce acute limb ischemia allowing for assessment of vascular regeneration, angiogenesis and arteriogenesis by a variety of strategies such as growth factor delivery and cell therapy. It can also be used to evaluate pro-angiogenic effects of therapeutic agents 3-12 or natural recovery in healthy 13 vs. diseased (diabetic) 14-16 young vs. aged 17, and wild type vs. knock out animals 18-20.
The PeriCam PSI Laser Speckle Contrast Imaging System is used to confirm ischemia post-surgery by measuring a decrease in blood perfusion in the microcirculation (60-70%) and longitudinally monitor the efficacy of the treatment strategy. Typically, each animal is followed for up to a month post-surgery/treatment with the PSI to document incremental increases in blood perfusion as a result of revascularization saving the researcher time and money by reducing the number of animals needed for a study.
Chicken Chorioallantoic Membrane (CAM) Assay
During the chicken embryo development, the CAM is formed by fusion of the mesodermal layer of the allantois with the mesodermal layer of the chorion. It is a highly vascularized, non-innervated extra embryonic membrane making it an ideal substrate for the study of angiogenesis and tumor growth. A fertilized chicken egg is incubated for four to eight days when a window is created in the shell to observe the forming embryo and blood vessels.
For the angiogenesis assay, various biomolecules and drugs can be topically delivered and studied for their angiogenic potency. Biomaterial scaffolds can also be implanted and monitored for vessel invasion overtime in order to assess a tissue engineering strategy.
For cancer models, various types of cancer cells can be transplanted into the CAM for tumor growth. This provides a relatively simple model for studying many different cancer tumor formations and allows for testing new therapy drugs and personalized treatment strategies. The CAM model is highly reproducible, cost effective, and does not require approval from an ethics board like many other in vivo animal models. Additionally, the CAM model has a natural immunodeficiency making it ideal for cell transplantation and the closed system prolongs the half-life of many experimental molecules, reducing the amount needed in studies.
The PeriCam PSI Laser Speckle Contrast Imaging System can be used to measure the change in blood perfusion in the angiogenesis assay and detect differences in the efficacy of various pro-angiogenic compounds. It can also be used to monitor the formation of functional intratumoral blood vessels 25 and assess the effectiveness of anti-angiogenic agents for cancer treatment.
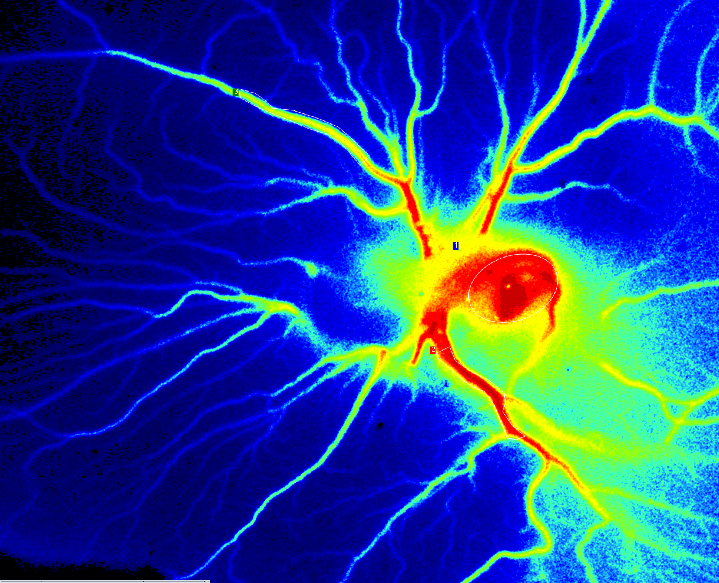
PSI Z perfusion of CAM Assay day 6 after fertilization.
Subcutaneous Tumor
The growth and metastasis of tumors depends on the development of blood vessels and an effective microcirculation. Without angiogenesis, a small focus of tumor cells cannot grow at a secondary site. Since angiogenesis plays a central role in tumor growth, progression, invasion, and metastasis, inhibiting it provides a potential strategy for some cancer treatment.
The Subcutaneous tumor model is the most popular assessment system for in vivo evaluation of novel anti-cancer medicine candidates. Usually using immunodeficient animal strains, cultured cancer cells are subcutaneously implanted resulting in solid tumor formation in about 2 weeks. A wide variety of human and murine cancer cell lines have been adapted to grow in a rodent host and thus allow evaluation of therapeutics in the relevant tumor model. Researchers monitor tumor growth and progression, and often perform a tumor microvessel density assay to assess the efficacy of their treatment strategy. The PeriCam PSI can be used to monitor the blood perfusion in the subcutaneous tumors over time and assess the efficacy of anti-angiogenic therapies for the treatment of cancer.
Perfusion images of mouse ear with subcutaneous tumor at day 14. Images reflect an increase in tumor blood perfusion in response to a heating challenge.
References:
1. LED-Based Photoacoustic Imaging for Monitoring Angiogenesis in Fibrin Scaffolds. Yunhao Zhu, Xiaofang Lu, Xiaoxiao Dong, Jie Yuan, Mario L. Fabiilli, and Xueding Wang. 9, September 2019, Tissue Engineering Part C: Methods, Vol. 25, pp. 523-531.
2. Controlled release of basic fibroblast growth factor for angiogenesis using acoustically-responsive scaffolds. Alexander Moncion, Melissa Lin, Eric G. O’Neill, Renny T. Franceschi, Oliver D. Kripfgans, Andrew J. Putnam, Mario L. Fabiilli. 2017, Biomaterials, Vol. 140, pp. 26-36.
3. MiR-15b-5p Regulates Collateral Artery Formation by Targeting AKT3 (Protein Kinase B-3). Ling-Ping Zhu, Ji-Peng Zhou, Jia-Xiong Zhang, Jun-Yao Wang, Zhen-Yu Wang, Miao Pan, Ling-. May 2017, Arteriosclerosis, Thrombosis, and Vascular Biology, Vol. 37, pp. 957-968.
4. Precise and long-term tracking of adipose-derived stem cells and their regenerative capacity via superb bright and stable organic nanodots. Dan Ding, Duo Mao, Kai Li, Xiaomin Wang, Wei Qin, Rongrong Liu, David Shunzhong Chiam, Nikodem Tomczak, Zhimou Yang, Ben Zhong Tang, Deling Kong, Bin Liu. 12, 2014, ACS Nano, Vol. 8, pp. 12620-12631.
5. Extracellular Matrix Hydrogel Promotes Tissue Remodeling, Arteriogenesis, and Perfusion in a Rat Hindlimb Ischemia Model. Jessica L. Ungerleider, Todd D. Johnson, Melissa J. Hernandez, Dean I. Elhag, Rebecca L. Braden, Monika Dzieciatkowska, Kent G. Osborn, Kirk C. Hansen, Ehtisham Mahmud and Karen L. Christman. 1-2, 2016, JACC: Basic to Translational Science, Vol. 1, pp. 32-44.
6. Engineering an Injectable Muscle-Specific Microenvironment for Improved Cell Delivery Using a Nanofibrous Extracellular Matrix Hydrogel. Nikhil Rao, Gillie Agmon, Matthew T. Tierney, Jessica L. Ungerleider, Rebecca L. Braden, Alessandra Sacco, Karen L. Christman. 2017, ACS Nano, Vol. 11, pp. 3851-3859.
7. Marrow-isolated adult multilineage inducible cells embedded within a biologically-inspired construct promote recovery in a mouse model of peripheral vascular disease. Cristina Grau-Monge, Gaëtan J-R Delcroix, Andrea Bonnin-Marquez, Mike Valdes, Ead Lewis Mazen Awadallah, Daniel F Quevedo, Maxime R Armour, Ramon B Montero, Paul C Schiller, Fotios M Andreopoulos and Gianluca D’Ippolito. 1, 2017, Biomedical Materials, Vol. 12, p. 015024.
8. Natural compound bavachalcone promotes the differentiation of endothelial progenitor cells and neovascularization through the RORα-erythropoietin-AMPK axis. Shuang Ling, Rong-Zhen Ni, Yunyun Yuan, Yan-Qi Dang, Qian-Mei Zhou, Shuang Liang, Fujiang Guo, Wei Feng, Yuanyuan Chen, Katsumi Ikeda, Yukio Yamori, and Jin-Wen Xu. 49, 2017, Oncotarget, Vol. 8, pp. 86188-86205.
9. Topical tissue nano-transfection mediates non-viral stroma reprogramming and rescue. Daniel Gallego-Perez, Durba Pal, Subhadip Ghatak, VeysiMalkoc, Natalia Higuita-Castro, Surya Gnyawali, Lingqian Chang, Wei-Ching Liao, Junfeng Shi, Mithun Sinha, Kanhaiya Singh, Erin teen, Alec Sunyecz, Richard Stewart, JordanMoore, Thomas Ziebro, Robert. 2017, Nature Nanotechnology, Vol. 12, pp. 974-979.
10. In vivo efficacy of endothelial growth medium stimulated mesenchymal stem cells derived from patients with critical limb ischemia. Rida Al-Rifai, Philippe Nguyen, Nicole Bouland, Christine Terryn, Lukshe Kanagaratnam, Gaël Poitevin, Caroline François, Catherine Boisson-Vidal, Marie-Antoinette Sevestre & Claire Tournois. 17, 2019, Journal of Translational Medicine, Vol. 9, p. 261.
11. NFAT5 promotes arteriogenesis via MCP‐1‐dependent monocyte recruitment. Zhang, Xing‐Chi Lin Miao Pan Ling‐Ping Zhu Quan Sun Zheng‐Shi Zhou Chuan‐Chang Li Guo‐Gang. 2, 2020, Journal of Cellular and Molecular Medicine, Vol. 24.
12. MicroRNA-146a Regulates Perfusion Recovery in Response to Arterial Occlusion via Arteriogenesis. Heuslein Joshua L., McDonnell Stephanie P., Song Ji, Annex Brian H., Price Richard J. 2018, Frontiers in Bioengineering and Biotechnology, Vol. 6, p. 1.
13. Denervation in Femoral Artery-Ligated Hindlimbs Diminishes Ischemic Recovery Primarily via Impaired Arteriogenesis. Yinghuan Cen, Junfeng Liu, Yuansen Qin, Ruiming Liu, Huijin Wang, Yu Zhou, Shenming Wang, Zuojun Hu. 5, 2016, PLOS One, Vol. 11, p. e0154941.
14. Elevating CXCR7 Improves Angiogenic Function of EPCs via Akt/GSK-3β/Fyn-Mediated Nrf2 Activation in Diabetic Limb Ischemia. Xiaozhen Dai, Xiaoqing Yan, Jun Zeng, Jing Chen, Yuehui Wang, Jun Chen, Yan Li, Michelle T Barati, Kupper A Wintergerst, Kejian Pan, Matthew A Nystoriak, Daniel J Conklin, Gregg Rokosh, Paul N Epstein, Xiaokun Li, Yi Tan. 2017, Circulation Research, Vol. 120, pp. e7-e23.
15. Sitagliptin-mediated preservation of endothelial progenitor cell function via augmenting autophagy enhances ischaemic angiogenesis in diabetes. Dai, X., Zeng, J., Yan, X., Lin, Q., Wang, K., Chen, J., Shen, F., Gu, X., Wang, Y., Chen, J., Pan, K., Cai, L., Wintergerst, K. A. and Tan, Y. 1, 2018, Journal of Cellular and Molecular Medicine, Vol. 22, pp. 89-100.
16. Endothelial Overexpression of Metallothionein Prevents Diabetes-Induced Impairment in Ischemia Angiogenesis Through Preservation of HIF-1α/SDF-1/VEGF Signaling in Endothelial Progenitor Cells. Kai Wang, Xiaozhen Dai, Junhong He, Xiaoqing Yan, Chengkui Yang, Xia Fan, Shiyue Sun, Jing Chen, Jianxiang Xu, Zhongbin Deng, Jiawei Fan, Xiaohuan Yuan, Hairong Liu, Edward C. Carlson, Feixia Shen, Kupper A. Wintergerst, Daniel J. Conklin, Paul N. Epstein. 8, August 2020, Diabetes, Vol. 69, pp. 1779-1792.
17. DNA Methyltransferase 1–Dependent DNA Hypermethylation Constrains Arteriogenesis by Augmenting Shear Stress Set Point. Joshua L. Heuslein, Catherine M. Gorick, Ji Song, Richard J. Price. 12, 2017, Journal of the American Heart Association, Vol. 6.
18. Loss of Endothelial CXCR7 Impairs Vascular Homeostasis and Cardiac Remodeling After Myocardial Infarction. Huifeng Hao, PhD, et al. 2017, Circulation, Vol. 135, pp. 1253-1264.
19. Vascular growth responses to chronic arterial occlusion are unaffected by myeloid specific focal adhesion kinase (FAK) deletion. Joshua L. Heuslein, Kelsey P. Murrell, Ryan J. Leiphart, Ryan A. Llewellyn, Joshua K. Meisner & Richard J. Price. 2016, Scientific Reports, Vol. 6, p. 27029.
20. Despite Normal Arteriogenic and Angiogenic Responses, Hindlimb Perfusion Recovery and Necrotic and Fibro-Adipose Tissue Clearance Are Impaired in MMP9 Deficient Mice. Meisner JK, Annex BH, Price RJ. 6, 2015, Journal of Vascular Surgery, Vol. 61, pp. 1583–1594.
21. Cutaneous Epithelial to Mesenchymal Transition Activator ZEB1 Regulates Wound Angiogenesis and Closure in a Glycemic Status–Dependent Manner. Singh, Kanhaiya and Sinha, Mithun and Pal, Durba and Tabasum, Saba and Gnyawali, Surya C. and Khona, Dolly and Sarkar, Subendu and Mohanty, Sujit K. and Soto-Gonzalez, Fidel and Khanna, Savita and Roy, Sashwati and Sen, Chandan K. 11, 2019, Diabetes, Vol. 68, pp. 2175-2190.
22. Correction of MFG-E8 Resolves Inflammation and Promotes Cutaneous Wound Healing in Diabetes. A. Das, S. Ghatak, M. Sinha, S. Chaffee, Noha S. Ahmed, N. L. Parinandi, E. S. Wohleb, J. F. Sheridan, C. K. Sen, and S. Roy. 12, 2016, The Journal of Immunology, Vol. 196, pp. 5089-5100.
23. Circulating Exosomal miR-20b-5p Inhibition Restores Wnt9b Signaling and Reverses Diabetes-Associated Impaired Wound Healing. Yuan Xiong, Lang Chen, Chenchen Yan, Wu Zhou, Yori Endo, Jing Liu, Liangcong Hu,Yiqiang Hu, Bobin Mi and Guohui Liu. 3, 2020, Small, Vol. 16, p. 1904044.
24. Saliva Exosomes-Derived UBE2O Promotes Angiogenesis in Cutaneous Wounds by Targeting SMAD6. Bobin Mi, Lang Chen, Yuan Xiong, Chenchen Yan, Hang Xue, Adriana C. Panay, Jing Liu, Liangcong Hu, Yiqiang Hu, Yun Sun, Faqi Cao, Wu Zhou, Guohui Liu. 18, 2020, Journal of Nanobiotechnology, Vol. 6, p. 68.
25. Laser speckle contrast analysis (LASCA) technology for the semiquantitative measurement of angiogenesis in in-ovo-tumor-model. Eric Pion, Claudia Asam, Anna-Lena Feder, Oliver Felthaus, Paul I. Heidekrueger,Lukas Prantl, Silke Haerteis, Thiha Aung. 2020, Microvascular Research, Vol. 133, p. 104072.
Please fill in the form to get more information
Disclaimer: It is possible that the products on the Perimed website may not be cleared for sale in all markets.