Recommended Products
PeriCam PSI NR

PeriFlux 6000 Combined System Stand Alone
PeriFlux 6000 EPOS

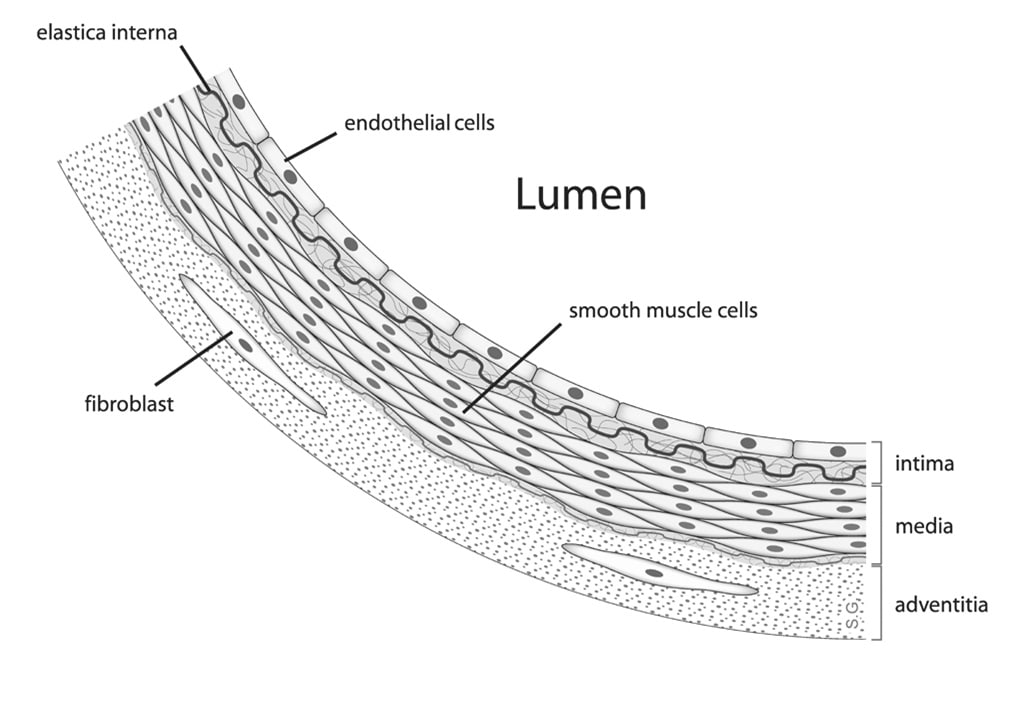
Endothelial Dysfunction
![]()
In several diseases, including prediabetes and diabetes, the vessels have a disturbed endothelium-dependent vasodilation and decreased bioavailability of nitric oxide (NO). This is called microvascular dysfunction or Endothelial Dysfunction. An increasing amount of evidence links Endothelial Dysfunction to cardiovascular outcomes: it can be a predictor for stroke and heart attacks, or risk for development of diabetes. The endothelial function can be studied by performing tests that cause endothelium-dependent and/or independent vasodilation. These tests include post occlusive reactive hyperemia (PORH), local heating (thermal challenge) and iontophoresis.
An increasing amount of evidence links microvascular dysfunction to cardiovascular outcomes: it can be a predictor for stroke and heart attacks. The endothelial function can be studied by performing tests that cause endothelium-dependent and/or independent vasodilation. These tests include post occlusive reactive hyperemia (PORH), local heating (thermal challenge) and iontophoresis.
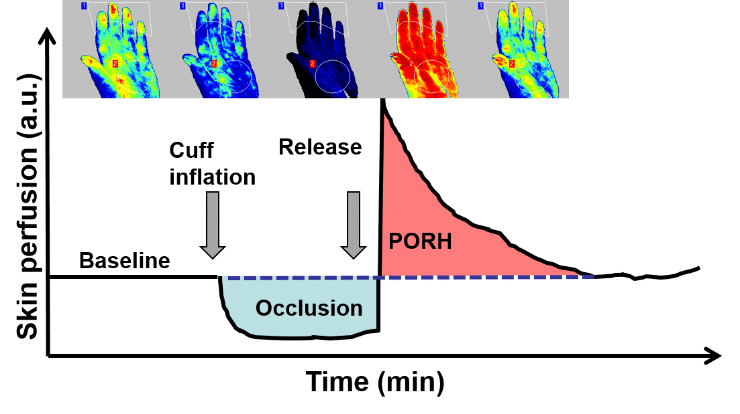
Post Occlusive Reactive Hyperemia (PORH)
Vascular function can be assessed by observing the response to reactive hyperemia. Reactive hyperemia is an increase in blood flow as a result of a temporary occlusion of an arterial blood supply leading to an oxygen deficit. Patients with impaired endothelial function show a different response than healthy controls. The test is done by performing an arterial occlusion, usually by using a blood pressure cuff, typically for several minutes. After the occlusion, the pressure is suddenly released, causing a large influx of blood into the previously occluded tissue. The perfusion is measured before, during and after the occlusion and several parameters related to both magnitude of the perfusion and the time it takes to change can give information about the vascular health of the patient.
The perfusion can be measured using a laser Doppler probe or a laser speckle contrast imager.
Read more about PORH
Thermal challenge/Heat provocation
Local heating of the skin may elicit near maximal vasodilatation. In healthy individuals the response to thermal hyperemia demonstrates a predictable pattern. It is characterized by an initial peak in skin blood perfusion, involving the sensory afferents, and a secondary rise and plateau mediated by NO release.
Read more about heat provocations.
The perfusion can be measured using a laser Doppler probe or a laser speckle contrast imager.
The heat can be controlled using a PeriFlux System 6000 with a heat control unit and a laser doppler probe with heat or a transparent water-filled heat probe for the imager.
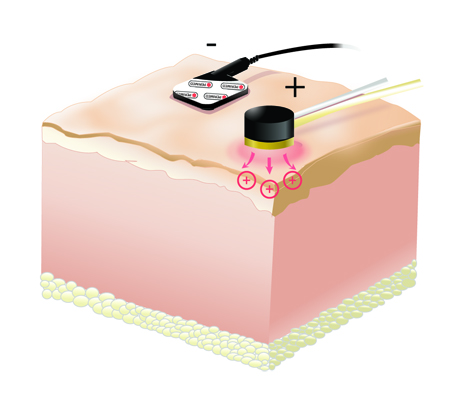
Iontophoresis
Iontophoresis is a technique to transport charged molecules or drugs across a tissue barrier. Combined with the laser Doppler or laser speckle techniques, iontophoresis is a valuable tool for diagnosis and studying endothelial dysfunction.
The perfusion can be measured by laser Doppler using a specialized delivery electrode with integrated laser Doppler probe or a laser speckle contrast imager, using a transparent delivery electrode for use with the imager.
Read more about Iontophoresis
References:
- Binggeli C. et al. Statins enhance postischemic hyperemia in the skin circulation of hypercholesterolemic patients. J Amer Collage of Card 2003 42; 1:71-77
- Ruano J. et al. Phenolic content of virgin olive oil improves ischemic reactive hyperemia in hypercholesterolemic patients. J Amer Collage of Card 2005 46; 10:1864-1868
- Roustit M. et al, Excellent reproducibility of laser speckle contrast imaging to assess skin microvascular reactivity. Microvascular Research, 2010
- Ann Humeau-Heurtier et al. Excellent inter- and intra-observer reproducibility of microvascular tests using laser speckle contrast imaging, Clinical Hemorheology and Microcirculation, 2013
- Roustit et al, Assessment of endothelial and neurovascular function in human skin microcirculation, Trends in Pharmacological Sciences, 2013, 34(7):373-84
- Kellogg D.L. Jr. In vivo mechanisms of cutaneous vasodilation and vasoconstriction in humans during thermoregulatory challenges. J Appl Physiol 2006;100:1709-1718.
- Gooding K.M. et al, Maximum Skin Hyperaemia Induced by Local Heating: Possible Mechanism. J Vasc Res 2006;43:270-277
- Minson C.T. et al, Nitric oxide and neurally mediated regulation of skin blood flow during local heating. J Appl Physiol 2001 91:1619-1626
- Minson C.T. et al, Thermal provocation to evaluate microvascular reactivity in human skin. J Appl Physiol 2010 109:1239-1246
- Blaauw J. et al. Abnormal endothelium-dependent microvascular reactivity in recently preeclamptic women. Amer Coll of Obstetricians and Gynecologists, 2005, 105; 3:626-632
- Durand S. et al. Prostaglandins participate in the late phase of the vascular response to acetylcholine iontophoresis in humans. J Physiol, 2004, 811-819
Case example:
The Microcirculation in Obesity and type 2 Diabetes
Dr. Alfons J.H.M. Houben Dept. of Internal Medicine, Maastricht University Medical Center+ and School for Cardiovascular Diseases (CARIM), Maastricht, The Netherlands
The focus of my research for many years has been microvascular dysfunction (MVD) as both a cause and a consequence of (cardio)metabolic diseases (see 1 for our working hypothesis). One important function of the microcirculation is to deliver oxygen/nutrients to all tissues and to remove waste products. In normal metabolism this includes the delivery of glucose, which taken up by the gut following a meal, to skeletal muscle in order to be stored as glycogen.
Please fill in the form to get more information
Disclaimer: It is possible that the products on the Perimed website may not be cleared for sale in all markets.