Change in medtech is inevitable — managing it ensures continuity for our customers
by Jimmy Bakker (Product Manager)
Recently, we upgraded the touchscreen interface of our perfusion monitor, PeriFlux 6000. While this may not sound revolutionary, the path from an end-of-life notification to a new component in production made me reflect on what it means to manage change in medtech.
As product manager, my responsibility is to ensure that our systems keep pace with the evolving needs of clinicians and researchers, advances in technology and materials, and an increasingly complex, changing regulatory and cybersecurity landscape.
In this post, I will share Perimed’s approach to change management — and how we design it to deliver the best outcomes for our customers, their patients, and our internal teams — using the touchscreen transitions as a practical example.

All change comes from necessity
There is always a driver. Sometimes it’s obvious: new clinical insights, updated regulations, or technological breakthroughs. Other times, the driver is quieter, almost invisible, like when a component reaches end-of-life.
This was the case for our touchscreen. The supplier informed us that the component we had been using would soon no longer be available — a clear signal to act. In a regulated environment like ours, such notifications are critical, giving us the lead time needed to plan and implement change in a structured way.
The touchscreen transition change became part of a broader hardware initiative, addressing additional capabilities — some of which have already been released: What’s new in PeriFlux 6000
Managing change holistically
A holistic approach allows us to avoid the pitfalls of managing change in isolation and uncover synergies. It enables us to remain agile while delivering updates in a controlled and reliable way.
When a component reaches end-of-life, the primary objective is to secure continuity of supply. From there, the key question becomes: do we implement a like-for-like replacement or take the opportunity to introduce a broader upgrade.
Even a seemingly straightforward replacement requires careful evaluation.
In medtech, this evaluation spans multiple dimensions, not least the materials used. For the touchscreen, we assessed:
- Material compliance — ensuring no hazardous or restricted substances are used.
- Biocompatibility — less critical here, but relevant, through indirect contact.
- Electrical safety — verifying leakage currents and updating inspection routines.
- Certification impact — aligning planned changes with regulatory compliance.
- Cleaning — resistance to repeated disinfection.
- Environmental resistance — performance under temperature, dust, and moisture variation.
- Tolerance and fit — integration and assembly precision.
- Signal integrity — no additional noise or interference.
- Supplier reliability and lead time — ensuring long-term availability and continuity of supply.
- Testing and validation — performance within the complete system.
- Cost — balancing new technology against current parts and assembly.
Regulatory considerations
Regulatory requirements are a major driver of change in medical technology. While they often evolve more slowly than hardware or software, their impact can be profound.
Maintaining compliance is essential for continued market access. Failure to do so can lead to product recalls, market restrictions, or, in extreme cases, inability to operate.
This is why we approach change holistically — considering not only current requirements, but also upcoming regulatory shifts as part of every decision.

User expectations
Clinicians increasingly expect the same level of responsiveness, simplicity, and ease of use as they experience in consumer technology.
Touchscreens are a clear example. From early adoption in industrial systems to widespread use in healthcare, they are now a standard in medical devices. Advanced technologies like eye tracking are enabling innovative interfaces, allowing users to control instruments hands-free — especially useful for surgical robots, where hands are busy.

The Perimed way
End-of-life events are often regarded as risks; at Perimed, we treat them as opportunities.
Replacing a component allows us to reassess original choices against current requirements and our broader development strategy. We evaluate durability, usability, and material standards, alongside internal considerations such as production efficiency, documentation, and serviceability.
Change isn’t purely about substitution; we approach it with a mindset of implementing meaningful and value-driven updates, while ensuring continuity.

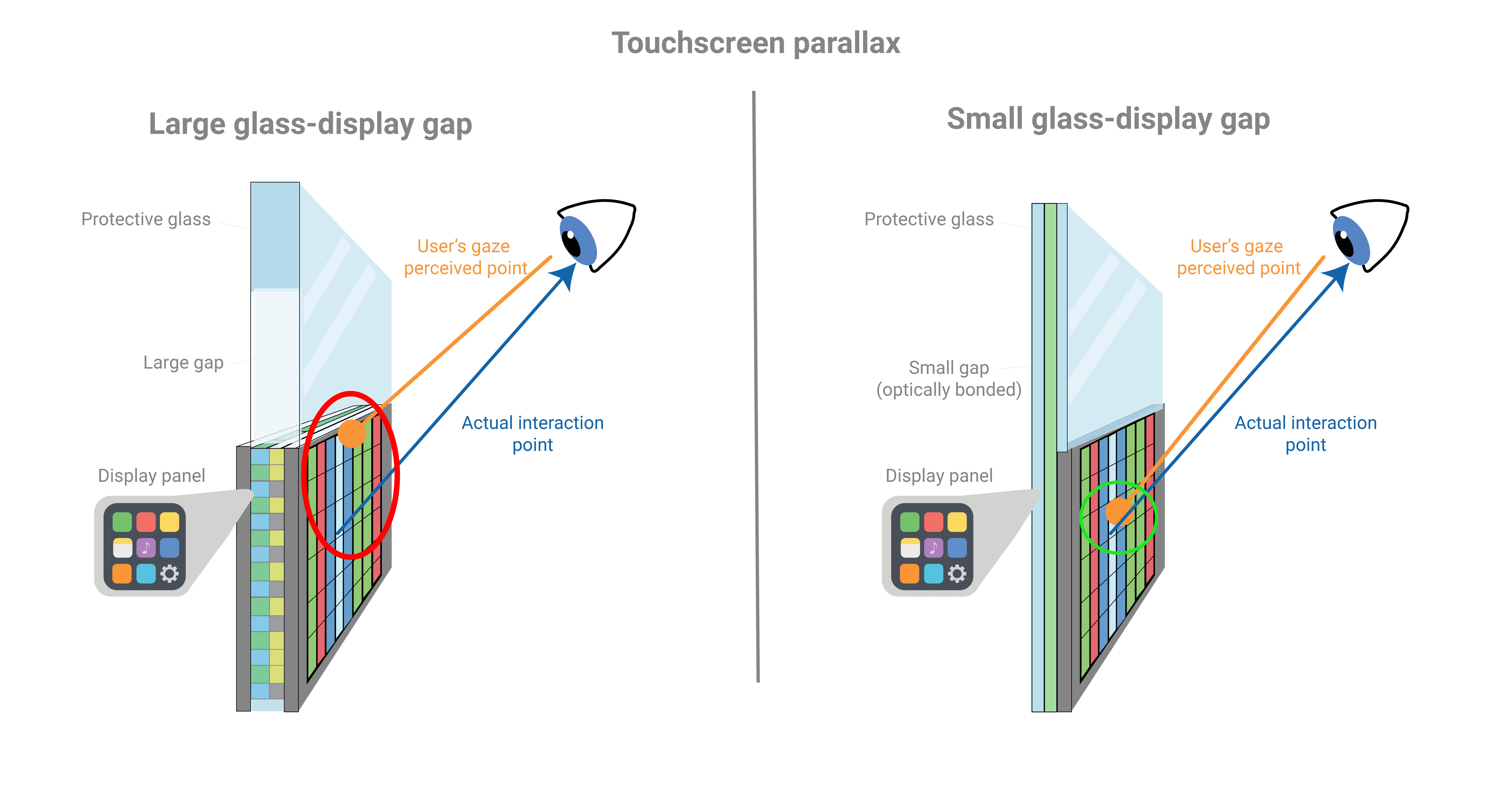
Our touchscreen — then and now
Then
In the early 2010s, it was standard engineering practice to deliver touch and display as separate components. While functional, the air gap between them introduces parallax — making the interface seem slightly offset from the point of touch.
The assembly was complex, involving around 20 discrete components, sourced from multiple suppliers. It required careful handling and testing in a controlled, dust-free environment.
Now
Advances in capacitive touch technology have fundamentally changed the capabilities. Our new touchscreen is clearer, more responsive, and fully integrated — eliminating parallax and reducing glare.
The number of components has significantly dropped, streamlining assembly and resulting in a more durable, maintainable, and serviceable solution that aligns with principles of resource efficiency and product sustainability.
For more information about PeriFlux 6000 and the range of macro- and microcirculation measurements it supports, please visit the product page.
Product Manager
I’ve been with Perimed for over 10 years, and in my role as PM for our imaging solutions, I get to work with amazing people who use our instruments to improve outcomes and find novel therapies for some of the world’s most prevalent diseases. I have a Master’s degree in applied physics and a Ph.D. in applied optics.

Disclaimer
The views expressed in this blog post are those of the author and do not necessarily represent the official position of Perimed. The content is provided for informational purposes only and is not intended to constitute clinical claims or medical advice. Clinical decisions should always be based on professional judgment and applicable clinical guidelines.