Understanding NICE HTG677 and what it means for healthcare in the UK
by Sara Gyllensporre (Clinical Application Specialist)
For more than a decade, I’ve been building relationships with clinicians and gaining valuable insights into how healthcare in the UK operates. As part of our strategy to scale in key markets, we recently partnered with HC 21 UK — one of Europe’s largest medtech providers. You can read more about that in this post: Strengthening vascular care across the UK.
On a recent visit to the UK, I was interested to learn that some hospitals I have worked closely with are no longer using certain medical devices listed in the NICE HTG677 recommendation, which recognizes the need for specialists to perform and interpret ankle brachial index (ABI) measurements. In short, the guidance concludes that more research is needed, and that the evidence is currently insufficient to support the routine use of automated ABI measurement devices to detect peripheral artery disease (PAD) in people with leg ulcers.
In this post, I will unpack what that means in practice. I will explore the two factors that affect ABI measurements: the physiological side of how underlying patient conditions can influence results, as well as the technical side, including how the equipment itself, regardless of method or instrument, can impact measurement quality. And lastly, I will share my thoughts on why — beyond patient outcomes — the recommendation exists and what it means for healthcare in the UK.

“For HC21 UK, NICE HTG677 highlights an opportunity to better support clinicians managing people with diabetes and complex wounds. By combining Perimed’s PeriFlux 6000 with our local service and education, we aim to help teams strengthen vascular assessment pathways and make more informed decisions about patient care.”
David Atkinson, Business Development Director — HC21 UK,
Leg ulcers and PAD
I think it’s important to put the recommendation into context. It specifically addresses the detection of peripheral artery disease (PAD) in people with leg ulcers. In other words, non-trivial diagnoses of complex patients with potential comorbidities.
As the recommendation explains, the rationale stems from the widespread use of compression therapy for leg ulcers. While effective, compression is not always an appropriate treatment. For individuals with underlying PAD, compression can further compromise blood flow to the limb, with potentially serious complications.
Why measure microcirculation
One of the positives with ABI is that it is a noninvasive way to assess arterial status, but it has its limitations. While low ABI is indicative of PAD, a normal ABI does not exclude it.
In some individuals, ABI may appear normal because blood can find alternative pathways around blockages. Arterial stiffness, which becomes more common with age and in people with diabetes or chronic kidney disease, can also affect the measurement. In these cases, ABI values may be falsely elevated because stiff arteries do not compress properly, causing ABI values to fall within the normal range despite underlying disease.
In isolation, ABI is not always reliable. It reflects large-vessel blood flow but does not capture tissue-level perfusion where healing occurs. Measuring microcirculation helps address this gap as it can reveal ischemia and provide deeper insights into healing potential and tissue viability.
Why then does the NICE recommendation focus on the technology rather than the inherent limitations of the measurement? My theory is the apparent simplicity of modern devices.
The shift to the home — at what cost?

The proliferation of devices for home- and community-based care is both inevitable and desirable. Driven by advances in technology and the growing need to support care at home and in remote settings, new devices are shifting diagnostics out of specialist environments and into settings closer to patients.
While this shift helps lower the burden on healthcare systems, it introduces a subtle challenge:
The assumption that ease of use equates to measurement reliability.
In the context of ABI, this assumption does not always hold.
Automated devices make it possible for almost anyone to take a measurement. The process appears simple: wrap a cuff around the limb, press a button, and read the result. However, alongside underlying physiological factors that can affect ABI accuracy, several additional parameters come into play, including:
- Correct cuff placement.
- Appropriate probe positioning.
- Interpretation of signal quality.
- Leaks in cuffs or tubing due to wear and tear.
- Limb positioning.
- Probe calibration.
All these factors require appropriate expertise, the right clinical setting, and suitable tools. I believe that this is what the NICE recommendation is pointing to in the following statement:
Explore the most appropriate user (specialist and non-specialist in assessing peripheral artery disease) and the most appropriate healthcare setting for their use.
Automated versus manual ABI
Is ABI the domain of specialist vascular clinics and hospital departments, staffed by trained personnel? Can it be reliably performed in primary care by a GP? Can patients carry it out at home with an automated device?
Manual ABI is typically performed using a handheld Doppler probe that converts arterial blood flow into an audible signal for the clinician to interpret.
This is the key point of the recommendation: automated ABI reduces, and maybe even eliminates, clinician involvement from the measurement process.
Keeping the clinician in the loop
To ensure that clinicians remain actively involved, Perimed has designed its ABI protocol to go beyond the generation of a single numerical value. Instead, it visualizes what’s happening throughout the entire measurement — from patient preparation, through baseline recording, occlusion, and finally the return of flow.
This continuous data capture provides insight into every phase of the measurement. It enables clinicians to assess not only whether the ABI is valid, but also whether the conditions under which it was obtained were valid, and identify physiological or technical factors that may be influencing the result.
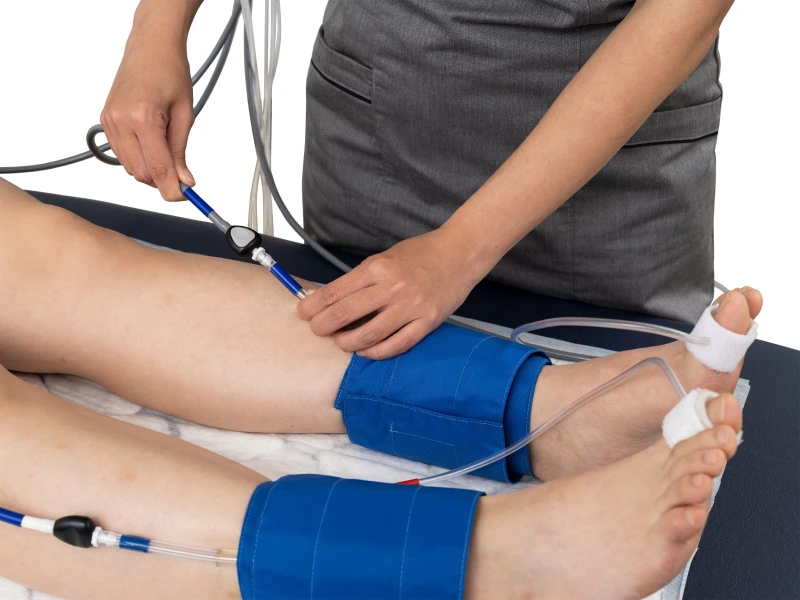
Figure (a) is a screenshot from our PeriFlux 6000 ABI protocol. It shows the baseline ankle pressure (area 1) before occlusion. This is not merely a starting point; it provides meaningful clinical context. For example, a PU below 20 may indicate underlying pathology such as microvascular disease or ischemia.
The thin gray line shows the pressure in the cuff, which rises rapidly as the cuff inflates and drops uniformly as the cuff deflates following occlusion. The system automatically identifies the point of return and records the corresponding pressure. However, this marker is not blindly accepted. Clinicians are expected to verify its accuracy, ensuring that manual interpretation remains central to the measurement.
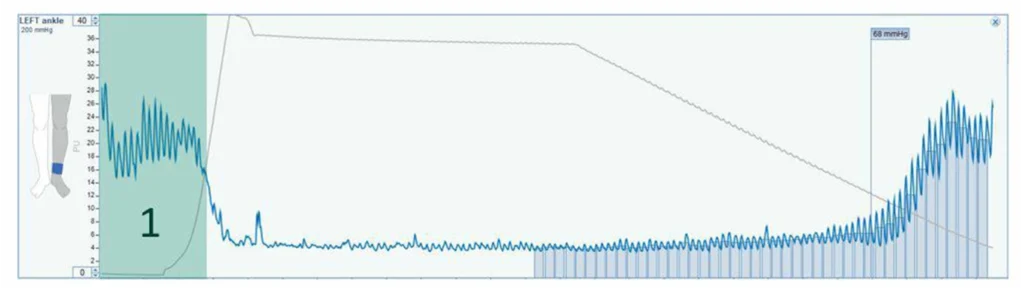
Figure (a) ABI measurement with PeriFlux 6000
In addition to the actual measurement, the details help clinicians to identify other issues, such as incompressible arteries and cuff wear and tear.
Signs of incompressible arteries include: pulsatility after occlusion, long occlusion times, or an ABI above 1.3 mmHg. The graph in Figure (b) indicates blood flow despite a cuff pressure of 202 mmHg.
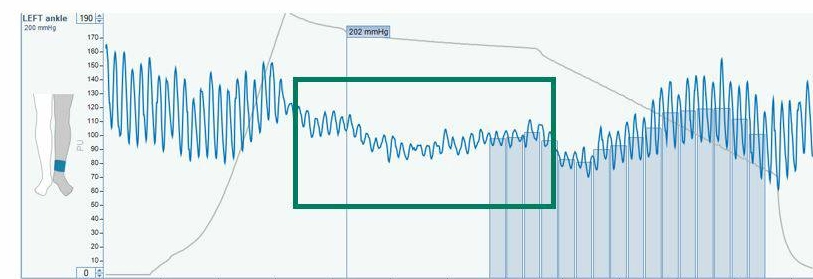
Figure (b) Signs of pulsatility
Our training material states that failure to achieve occlusion at 200 mmHg may indicate high blood pressure, arterial calcification, or other vascular abnormality. In such cases, we recommend repeating the measurement at a slightly higher pressure, 15-20 mmHg above the standard 200 mmHg, or proceeding with a toe pressure measurement.
The advantage of PeriFlux 6000 over ABI-only solutions is that it supports a comprehensive range of assessments, providing clinicians with insight into tissue viability.
- Macrovascular — including ABI, Exercise ABI, TBI, and PVR
- Microvascular — skin perfusion pressure (SPP), TcpO2, and Exercise TcpO2
Technical limitations, irrespective of method or instrument
Over time, pressure cuffs can wear out, and issues can arise with the connections and tubing between the air outlet and the cuff. These technical factors can affect measurement accuracy regardless of whether the system is manual or automated.
A non-linear cuff-pressure curve, as shown in the reference diagram, indicates an air leak, which needs to be corrected to ensure a valid ABI reading.
What it means for healthcare in the UK
The push to decentralize vascular assessment is understandable. While NICE HTG677 challenges the assumption that ABI is straightforward, it’s not a blanket rejection of automated ABI in favor of traditional manual methods. Rather, it can be read as a caution against deploying systems that oversimplify the measurement and diminish the importance of operator know-how and specialist vascular expertise. Without these, the risk is clear: convenience may come at the expense of diagnostic confidence.
I hope you found this post insightful. To find out more about ABI with PeriFlux 6000 visit: Ankle pressure and ABI.
Clinical Application Specialist
With over 16 years at Perimed, Sara has held multiple roles and developed a broad understanding of medtech and its application in healthcare settings. She holds a degree in pharmacology and an MSc in molecular biology and plays a vital role ensuring technology meets the needs of clinical practice, supported by strong relationships with clinicians across Europe.

Disclaimer
The views expressed in this blog post are those of the author and do not necessarily represent the official position of Perimed. The content is provided for informational purposes only and is not intended to constitute clinical claims or medical advice. Clinical decisions should always be based on professional judgment and applicable clinical guidelines.